|
4/21/2024 0 Comments Valence electron periodic table 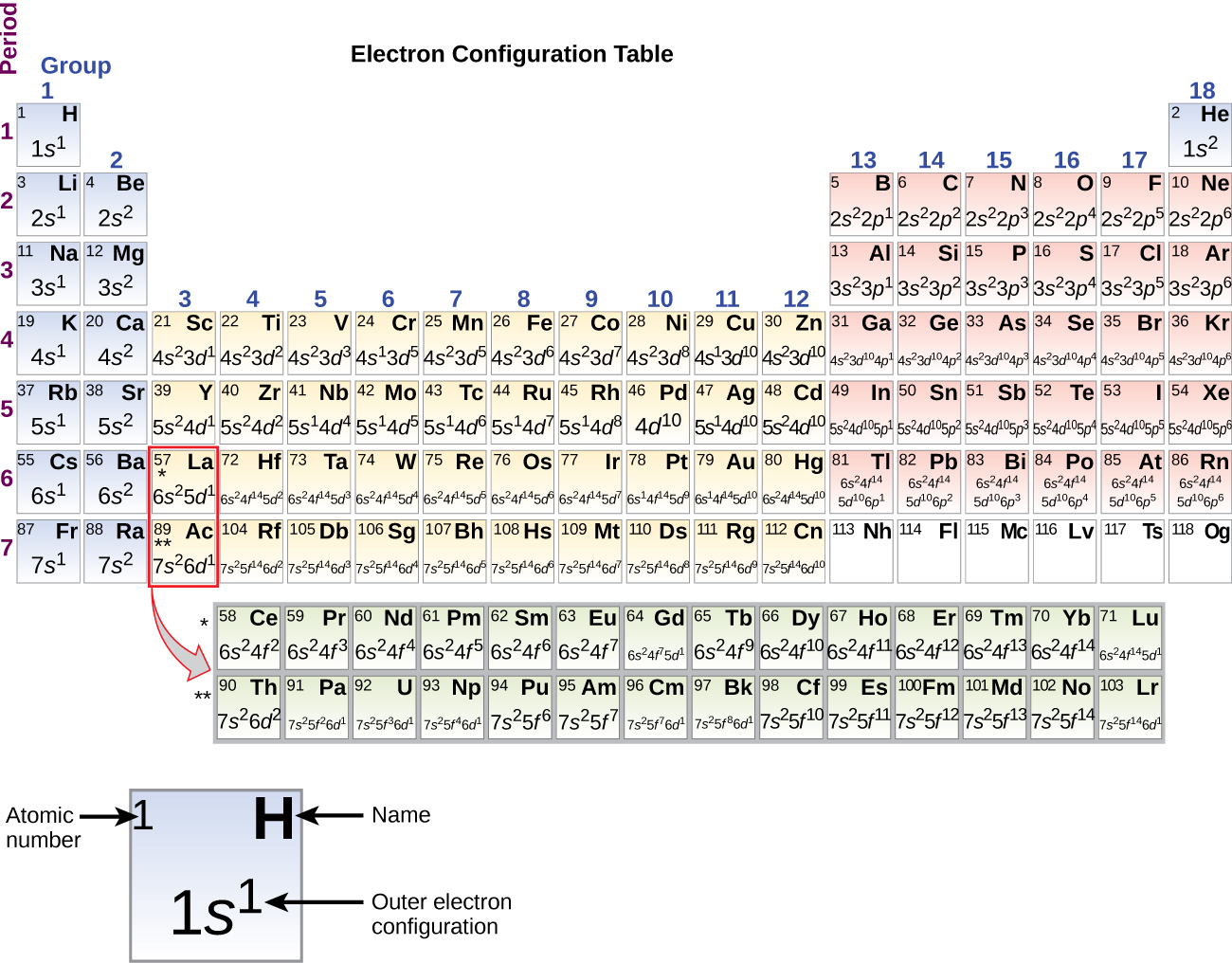
When an electron loses energy (thereby causing a photon to be emitted), then it can move to an inner shell which is not fully occupied. Or the electron can even break free from its associated atom's valence shell this is ionization to form a positive ion. An energy gain can trigger an electron to move (jump) to an outer shell this is known as atomic excitation. Like an electron in an inner shell, a valence electron has the ability to absorb or release energy in the form of a photon. An atom with one or two valence electrons fewer than a closed shell is also highly reactive, because of a tendency either to gain the missing valence electrons (thereby forming a negative ion), or to share valence electrons (thereby forming a covalent bond). An atom with one or two valence electrons more than a closed shell is highly reactive, because the extra valence electrons are easily removed to form a positive ion. The presence of valence electrons can determine the element's chemical properties and whether it may bond with other elements: For a main group element, a valence electron can only be in the outermost electron shell.Īn atom with a closed shell of valence electrons (corresponding to an electron configuration \(s^2p^6\)) tends to be chemically inert. Without knowing the valence electrons, we cannot draw Lewis structures which means we cannot determine the geometry and hybridization of the atom.\)Ī valence electron is an electron that is associated with an atom, and that can participate in the formation of a chemical bond in a single covalent bond, both atoms in the bond contribute one valence electron in order to form a shared pair. This is because they have the same valence electron configuration just different energy levels. Remember from the electron configurations that for transition metals, the (n+1)s energy level is filled before the (n)d level.īecause valence electrons are the farthest from the nucleus, they are the ones that are not bound as strongly and thus participate in chemical bonding and define the properties of the element.Įlements in the same group have similar chemical and physical properties. For example, iron has eight valence electrons: Fe – 1s 22s 22p 63s 23p 6 4s 23d 6, titanium has four electrons – Ti – 1s 22s 22p 63s 23p 6 4s 23d 2. If it is noted as 14 instead of 4A, you can subtract 10 from the group number and determine the valence electrons.įor d block elements, the outermost d electrons are also counted as valence electrons (ns + (n-1)d). This notation when the element is shown surrounded by its valence electrons is called Lewis dot symbols, and it is used for drawing the Lewis structure of molecules. For example, Mg is a Group 2A element and has two dots for two valence electrons, and C has four dots in the Lewis symbol since it is in Group 4A, and oxygen has six valence electrons: The number of valence electrons, for main group elements, corresponds to their group number in the periodic table. The rest of the electrons in the lower energy levels are called core electrons. 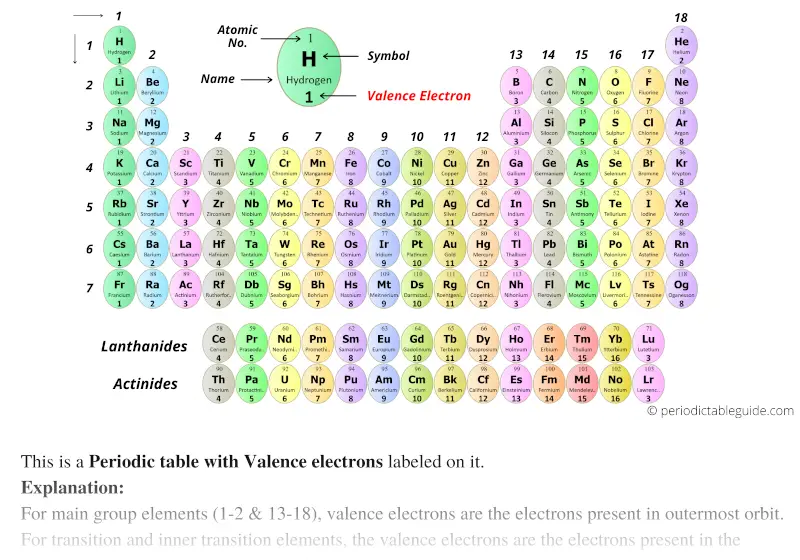
For example, sodium has one valence electron, and chlorine has seven valence electrons: Na – 1s 22s 22p 6 3s 1, Cl – 1s 22s 22p 6 3s 23p 5. Valence electrons are those in the outermost principal energy level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
